Therapeutic Antibody Development
Advance discovery by accelerating screening and optimization
Shatter workflow bottlenecks with automated benchtop solutions that propel discovery. Telesis Bio’s comprehensive automated synthetic biology solutions enable antibody therapeutic developers to rapidly build DNA constructs throughout lead candidate discovery and optimization workflows.
With benchtop automation solutions that enable push button synthetic biology and streamline multiomic applications in your lab, you can take control of your workflow and expedite the time to novel, high-impact discoveries.
Synthesis challenges slow antibody discovery
If your antibody discovery program is limited by the speed of your capabilities in synthesis and cloning of lead candidates, you’re not alone.
Identifying high-affinity antibodies against difficult target classes requires scientists to regularly generate, build and screen large numbers of leads candidates.
At Telesis Bio, we understand that to meet this challenge, researchers currently depend upon variable service provider deliveries or labor-intensive manual benchtop protocols to meet demanding project milestones.
But what if it was possible to build antibody sequence constructs hands-free overnight?
Break through to a new pace of discovery
The BioXp® automation platform streamlines screening and optimization workflows of antibody candidates by enabling high-throughput DNA synthesis overnight, and at the push of a button.
Overnight synthesis
On your own schedule
In your own lab
Hands-free automation

Variant Libraries
- High-diversity libraries for display panning
- Mutate variable regions for affinity maturation

DNA Amplification
- Transfection-scale DNA in hours
- Cell-free amplifications
- No bacterial culture required
How Tavotek leveraged automated synthetic biology solutions to reduce critical lead synthesis bottlenecks by 66% and to accelerate antibody discovery timelines to only 6 weeks
Case Study: Like many other antibody discovery programs, Tavotek felt constrained by the speed of their synthesis and cloning of lead candidates. By harnessing recent advances in automated synthetic biology from Telesis Bio, they have built a much more efficient lead discovery solution. One that delivers the speed and efficiency truly required in discovery workflows for complex antibody-based drugs and therapeutics.
Applications of Telesis Bio Synthetic Biology Solutions in Therapeutic Antibody Development

Digital Antibody Sequence to Transfection-ready Material in a Single Run
Infographic Brochure: What if you could synthesize lead candidate variable domains, clone them into your expression vectors, and amplify the resulting plasmids to transfection scale with a single push of a button?
Download this brochure to learn about how Telesis Bio’s synthetic biology solutions and automated BioXp workstation accelerate antibody discovery workflows
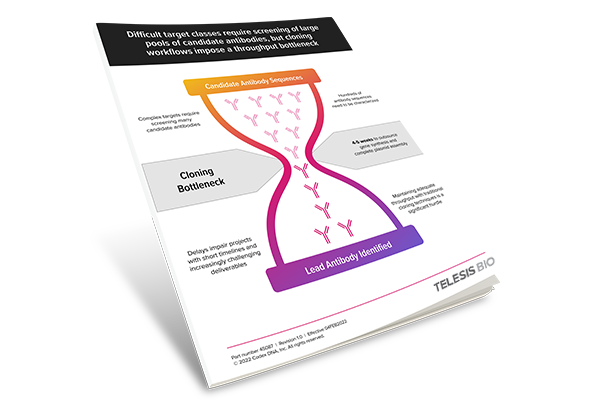

Bridging the Throughput Gap: Process Optimization in Antibody Discovery Workflows
White paper: Learn how process optimization utilizing automated synthetic biology solutions can remove the antibody sequence cloning bottleneck that divides high-content lead identification and downstream high throughput characterization.
Rethinking Lead Optimization: Adopting New Synthetic Biology Tools to Accelerate Development Timelines
White paper: Learn how advances in synthetic biology automation are enabling researchers to streamline antibody discovery and optimization workflows



